Medical Device Contract Manufacturing Market Scope and Industry Outlook to 2031
Medical Device Contract Manufacturing Market Scope
The medical device contract manufacturing industry plays a crucial role in supporting the global healthcare technology ecosystem. The Medical Device Contract Manufacturing Market Scope covers a wide range of manufacturing services, medical device technologies, packaging solutions, and regulatory classifications that collectively shape the industry. Contract manufacturing organizations support medical device companies by providing expertise in product design, engineering, production, assembly, and packaging while ensuring strict regulatory compliance.
The scope of the industry extends across the entire lifecycle of medical device production. Contract manufacturers assist in early stage product development, prototype testing, large scale manufacturing, and final packaging. These services enable medical device companies to accelerate product commercialization while maintaining high quality standards and meeting global regulatory requirements. As medical technologies become more sophisticated, the role of contract manufacturers continues to expand within the healthcare supply chain.
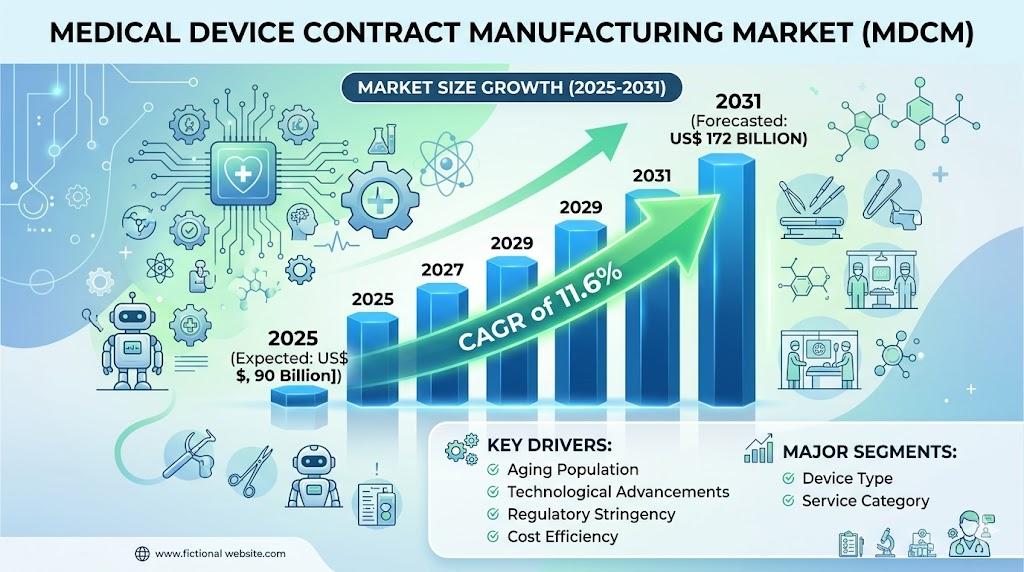
The industry is expected to witness strong growth in the coming years. The market was valued at approximately US$ 79.78 billion in 2024 and is projected to reach nearly US$ 172 billion by 2031, growing at a compound annual growth rate of 11.6 percent between 2025 and 2031. This growth reflects increasing demand for advanced medical devices, the rising complexity of healthcare technologies, and the growing reliance on outsourcing strategies among medical device companies.
Scope by Technology
The scope of the contract manufacturing sector includes a diverse range of medical device technologies used in modern healthcare systems. Contract manufacturers produce components and complete devices for several specialized medical fields.
Major technology areas within the industry include:
-
In vitro diagnostic devices
-
Diagnostic imaging devices
-
Cardiovascular devices
-
Drug delivery devices
-
Orthopedic devices
-
Ophthalmology devices
-
Diabetes care devices
-
Dental devices
-
Endoscopy devices
-
Respiratory care devices
-
Surgical devices
-
Gynecology and urology devices
-
Neurology devices
-
Personal care devices
-
Other medical technologies
These technology segments demonstrate the wide manufacturing capabilities required to support both simple diagnostic devices and highly complex implantable technologies.
Download Sample PDF : https://www.theinsightpartners.com/sample/TIPRE00006532
Scope by Service Type
The market scope also includes a broad range of services provided by contract manufacturing organizations. These services help medical device companies manage different stages of product development and commercialization.
The primary service segments include:
-
Device development and manufacturing services
-
Quality management services
Device development and manufacturing services focus on product design, engineering, prototyping, testing, and large scale production. Quality management services ensure that devices meet strict regulatory standards through quality assurance processes, regulatory documentation, and compliance monitoring.
Scope by Packaging and Assembly
Packaging and assembly services represent another important aspect of the contract manufacturing industry. Medical devices must be carefully packaged to maintain sterility, ensure product safety, and meet regulatory labeling requirements.
Packaging and assembly services typically include:
-
Primary and secondary packaging
-
Product labeling and regulatory identification
-
Specialized packaging solutions for transportation and storage
Proper packaging ensures that medical devices remain protected during distribution while maintaining their functional integrity and safety.
Scope by Device Classification
Medical devices produced through contract manufacturing are classified into different regulatory categories based on risk levels. These classifications determine the level of testing, regulatory review, and compliance required before products can enter the healthcare market.
The three main device classifications include:
-
Class I Medical Devices representing low risk devices such as basic surgical tools and monitoring equipment
-
Class II Medical Devices involving moderate risk technologies including diagnostic and therapeutic devices
-
Class III Medical Devices representing high risk devices such as implantable cardiovascular equipment and life supporting technologies
Contract manufacturing organizations must maintain advanced quality control systems and regulatory expertise to support production across these device classes.
Geographic Scope of the Industry
The global scope of the medical device contract manufacturing sector extends across several major regions. These regions contribute to industry development through manufacturing capabilities, technological innovation, and healthcare demand.
Key regions covered within the market scope include:
-
North America
-
Europe
-
Asia Pacific
-
South and Central America
-
Middle East and Africa
North America remains one of the leading regions due to strong healthcare infrastructure and the presence of major medical device manufacturers. Europe also represents a significant market supported by advanced medical technology development. Meanwhile, Asia Pacific is emerging as a rapidly growing region due to expanding healthcare demand, cost effective manufacturing capabilities, and increasing government investments in medical technology.
Future Outlook
The scope of the medical device contract manufacturing industry is expected to expand further as healthcare technologies continue to evolve. Increasing demand for advanced diagnostic tools, minimally invasive surgical devices, and personalized medical technologies will drive the need for specialized manufacturing capabilities. Contract manufacturing organizations are likely to invest in automation, digital manufacturing systems, and advanced quality management platforms to support these emerging healthcare innovations. As the global healthcare sector continues to grow, the scope of contract manufacturing services will remain a key factor supporting the development and production of next generation medical devices worldwide.
Trending Reports :
Skin Cancer Diagnostics Market
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Information -
Email: [email protected]
Phone: +1-646-491-9876
Also Available in : Korean| German| Japanese| French| Chinese| Italian| Spanish

- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jogos
- Gardening
- Health
- Início
- Literature
- Music
- Networking
- Outro
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


